Remote auditing (or e-auditing) is the practice of conducting compliance and quality audits entirely via digital tools, without auditors needing to be on-site. Auditors and clients meet in a virtual environment (for example via Microsoft Teams or Zoom) to share documents, interview staff, and review processes in real time. This method became widely adopted during
Read MoreHere’s a scenario that plays out more often than anyone in the pharmaceutical industry likes to admit. A pharmaceutical audit is scheduled for five days. Then the site has a shutdown. A key SME goes on leave. The client pushes for an early closeout. Suddenly you are doing a five-day audit in three — and
Read MoreIn 2026, internal audit teams face complex, fast-changing risks. A risk-based auditing approach ensures audits focus on an organization’s highest threats and objectives. This modern method aligns audit planning with enterprise risk management and strategic goals. It moves beyond box-checking to prioritize areas where failures could derail the business. By leveraging tools like AI and
Read MoreGetting FDA compliance wrong is expensive. A single audit observation can delay product releases, trigger warning letters, or shut down operations entirely. A computer system validation flow chart removes that uncertainty. It gives your quality, IT, and regulatory teams a shared, documented roadmap — from system planning all the way through retirement. This guide covers
Read MoreIn today’s digital-first life sciences environment, pharmaceutical, biotech, and medical device companies face relentless regulatory scrutiny. Agencies like the FDA and EMA require firms to demonstrate continuous control of their Good Practice (GxP) processes to ensure product quality, data integrity, and patient safety. Current Good Manufacturing Practice (CGMP) regulations set minimum standards for manufacturing methods
Read MoreIn today’s highly regulated life sciences landscape—encompassing pharmaceuticals, biotechnology, and medical devices—the reliability of computer systems is not merely a technical concern but a fundamental patient safety imperative. Computer System Validation (CSV) serves as the critical bridge between advanced technology and stringent regulatory compliance, ensuring that every system performs its intended function consistently and reliably.
Read MoreIn the high-stakes world of pharmaceuticals, biotechnology, and medical devices, the importance of the validation of the system cannot be overstated. It is the fundamental, non-negotiable process that stands between a functional software application and a trustworthy, compliant asset that protects patient lives and ensures product quality. Understanding the importance of the validation of the
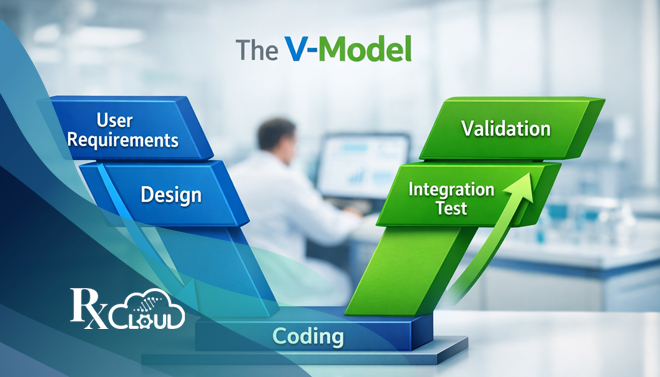
Read MoreIn the highly regulated landscape of life sciences—where a single software flaw can impact patient safety and regulatory approval—the validation of computer systems is not just best practice; it’s an absolute mandate. For pharmaceutical, biotechnology, and medical device companies, navigating the stringent requirements of Good Practice (GxP) regulations demands a robust, structured, and defensible methodology.
Read MoreIntroduction to GCP Audit In the evolving and highly regulated clinical research environment, maintaining compliance with Good Clinical Practice (GCP) is essential. A GCP Audit (Good Clinical Practice Audit) is a structured and independent evaluation of clinical trial activities to ensure that studies are conducted ethically, safely, and in accordance with regulatory requirements. Clinical trials
Read MoreGVP modules form the cornerstone of modern pharmaceutical safety monitoring in the European Union. According to the World Health Organization (WHO), pharmacovigilance is defined as “The science and activities relating to the detection, assessment, understanding, and prevention of adverse effects or any other drug-related problem”. We recognize how crucial these practices are for ensuring that
Read More